In microbiology, microorganisms are too small to be seen individually without specialized tools, yet understanding how many are present in a sample is essential. Measuring microbial growth helps scientists determine the severity of infections, monitor contamination in water and food, and evaluate the effectiveness of antimicrobial treatments. Because bacteria multiply rapidly and often exist in large populations, microbiologists rely on several laboratory methods to estimate their numbers. These methods can be grouped into direct approaches that count cells and indirect approaches that measure properties related to cell presence or activity.
Direct Microscopic Count Using the Petroff Hausser Chamber
The direct microscopic count is one of the most straightforward laboratory methods used for measuring microbial growth because it allows microbiologists to visually count bacterial cells in a known volume of liquid culture. This method uses a special calibrated counting slide known as the Petroff Hausser chamber, which is designed with a precisely etched grid to help estimate microbial concentration.
Step 1: Preparing the microbial suspension
A small sample of the bacterial culture is first mixed well to ensure that cells are evenly distributed. This is important because clumped cells can lead to inaccurate estimates of population size. If the culture is very dense or highly turbid, it may be diluted with sterile fluid so that individual cells can be seen clearly under the microscope.
Step 2: Loading the Petroff Hausser chamber
A measured drop of the suspension is carefully placed onto the chamber and covered with a coverslip. The coverslip sits at a fixed height above the grid surface, creating a known microscopic volume. This controlled volume is what makes it possible to calculate the number of cells per milliliter later. Proper loading is essential to avoid air bubbles or uneven distribution of cells.
Step 3: Observing the grid under the light microscope
The slide is then examined using a light microscope. The etched grid divides the viewing area into multiple squares of known dimensions. Microbiologists focus on selected squares and count the number of visible bacterial cells within those boundaries. This systematic counting reduces bias and improves accuracy when estimating microbial growth.
Step 4: Calculating cell concentration
Because both the area of each square and the depth of the chamber are known, the volume represented by each counted region can be determined. The average number of cells counted in several squares is then used in a mathematical calculation to estimate the total number of cells present in one milliliter of the original culture. This allows scientists to quantify microbial population density even though the organisms themselves are microscopic.
Step 5: Interpreting results and recognizing limitations
Although this method provides a rapid estimate of total microbial cell number, it does not automatically distinguish between viable and nonviable cells. Special fluorescent stains may be used when differentiation between live and dead cells is required. Additionally, very high cell densities can make counting difficult, which is why dilution is sometimes necessary. Despite these limitations, the Petroff Hausser chamber remains an important teaching and research tool because it clearly illustrates how microbiologists measure microbial growth directly.
Read Also: Neurotropic Mechanisms in Rabies Pathogenesis
Plate Count and the Concept of Colony Forming Units
The plate count method, also known as the viable plate count, is a key technique used in microbiology for measuring microbial growth because it estimates only the number of living bacteria that are capable of reproduction. This method allows microbiologists to determine the size of a viable microbial population by observing colony formation on solid culture media.
Step 1: Preparing the sample for plating
A bacterial culture or environmental sample is first prepared, and in many cases it is diluted to reduce the number of microorganisms present. This step is important because microbial populations can be extremely dense, making accurate counting impossible if too many cells are placed on one plate. Proper sample preparation ensures that individual cells are well separated when transferred to the growth medium.
Step 2: Inoculating the agar plate
A measured volume of the prepared sample is then transferred onto a sterile nutrient agar plate. This can be done using the spread plate method, where the sample is evenly distributed across the surface of solid agar, or the pour plate method, where the sample is mixed with warm liquid agar before it solidifies. Both approaches aim to isolate cells so that they can grow into separate colonies.
Step 3: Incubating to allow colony formation
The inoculated plates are placed in an incubator under suitable environmental conditions such as the correct temperature and oxygen availability. During incubation, each viable bacterial cell divides repeatedly through binary fission and produces a visible mass of cells known as a colony. These colonies represent successful microbial growth on the plate.
Step 4: Counting colonies and applying the CFU concept
After incubation, microbiologists count the number of colonies present on plates that contain a countable range of growth. Because it is not always certain whether a colony developed from one cell or a small cluster of cells, results are expressed as colony forming units per milliliter, abbreviated as CFU per milliliter. This unit reflects the number of viable microorganisms in the original sample that were capable of forming colonies.
Step 5: Selecting statistically reliable plates
For accurate estimation of microbial growth, plates with between thirty and three hundred colonies are typically used. Plates with fewer than thirty colonies may not provide reliable statistical representation of the population, while plates with more than three hundred colonies often show overlapping growth that makes individual counting difficult. This careful selection improves precision in microbial quantification.
Step 6: Interpreting the importance of viable counts
The plate count method is especially valuable because it measures living cells, which are the most important in clinical microbiology, food safety testing, environmental monitoring, and industrial quality control. By focusing on viable organisms, microbiologists can better assess infection severity, contamination risk, and the effectiveness of antimicrobial treatments.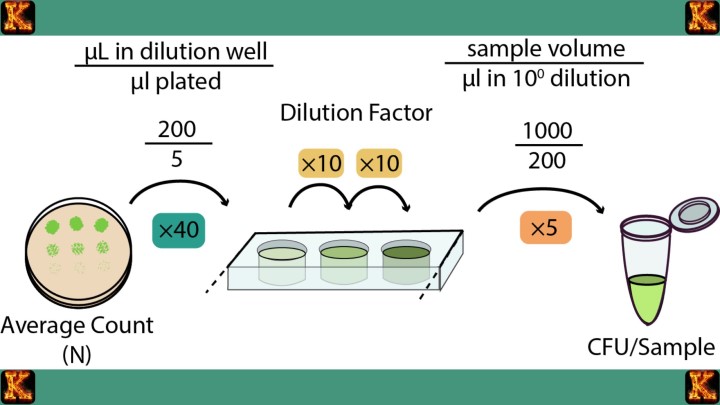
Serial Dilution Logic in Microbial Counting
Serial dilution is an essential laboratory technique used in measuring microbial growth because it makes it possible to handle extremely large bacterial populations in a controlled and logical way. Since microbial cultures can contain millions or even billions of cells per milliliter, plating the original sample directly would result in overcrowded growth that cannot be counted accurately. Serial dilution solves this problem by reducing cell concentration step by step.
Step 1: Preparing the original microbial sample
The process begins with a well mixed microbial culture to ensure that cells are evenly distributed throughout the suspension. This step is important because uneven distribution can lead to inaccurate estimates of microbial population size. A sterile dilution fluid such as buffered saline or nutrient broth is prepared in advance to maintain cell stability during dilution.
Step 2: Performing the first dilution
A fixed and measured volume of the original culture is transferred into a tube containing a larger volume of sterile dilution fluid. This commonly produces a tenfold dilution, meaning the concentration of microorganisms is reduced to one tenth of the original level. Proper mixing after transfer ensures that cells are uniformly dispersed within the new dilution.
Step 3: Creating a dilution series
A portion of the first diluted sample is then transferred into another tube of sterile fluid to produce the next tenfold dilution. This step is repeated several times to generate a serial dilution series with progressively lower microbial concentrations. Each dilution step follows a predictable mathematical pattern, which is why serial dilution is described as systematic and exponential.
Step 4: Plating selected dilutions for colony counting
Measured volumes from one or more dilution tubes are inoculated onto agar plates using methods such as spread plating or pour plating. After incubation, colonies form from viable cells. Because the dilution process reduces cell density, at least one plate is expected to contain a countable number of colonies, which is necessary for reliable estimation of microbial growth.
Step 5: Calculating the original microbial population
Once colonies are counted, the dilution factor is applied in a mathematical calculation to estimate the number of microorganisms present in the original undiluted culture. This step allows microbiologists to convert colony counts into meaningful values such as colony forming units per milliliter. Through this logic, serial dilution makes it possible to quantify very large microbial populations with accuracy and confidence.
Read Also: How Microscopy Helps Identify Bacteria in Clinical Samples
Most Probable Number Method
The Most Probable Number method, abbreviated as MPN, is a statistical approach used in measuring microbial growth when direct counting methods are not practical. This technique is especially useful for estimating the number of viable microorganisms in samples such as drinking water, food products, or environmental sources where bacterial populations may be low or unevenly distributed.
Step 1: Preparing sets of broth tubes
The procedure begins by arranging several sets of sterile broth tubes that contain a suitable growth medium. In many applications, the tubes contain lactose broth with a pH indicator, which helps detect microbial metabolic activity. These sets are designed to receive different volumes of the test sample so that microbial growth can be observed across a range of concentrations.
Step 2: Inoculating tubes with different sample volumes
Measured volumes of the microbial sample are added to each set of tubes. Typically, larger volumes are inoculated into one set, moderate volumes into another, and very small volumes into a third set. This structured inoculation creates varying probabilities of microbial presence in each tube, which forms the basis of the statistical reasoning used in the MPN method.
Step 3: Incubating to allow microbial growth and fermentation
The inoculated tubes are incubated under appropriate environmental conditions. If coliform bacteria or other lactose fermenting microorganisms are present, they metabolize lactose and produce acidic byproducts. This metabolic activity causes the pH indicator to change color, often from red to yellow, signaling a positive growth response.
Step 4: Recording positive and negative results
After incubation, microbiologists examine each tube and record whether growth has occurred. Tubes that show turbidity, gas production, or color change are considered positive, while those that show no visible changes are considered negative. The overall pattern of positive and negative tubes across the dilution sets provides important information about microbial distribution in the sample.
Step 5: Estimating microbial concentration using probability tables
The recorded pattern is then compared with standardized MPN probability tables, which are used to estimate the most likely concentration of viable microorganisms in the original sample. Although this method does not give an exact numerical count, it provides a scientifically valid estimate of microbial population size. This makes the MPN method highly valuable in situations where other techniques are limited by low cell numbers, particulate matter, or sample turbidity.
Spectrophotometry and Indirect Measurement of Microbial Growth
Spectrophotometry is an important indirect method for measuring microbial growth because it estimates bacterial population size without counting individual cells. Instead of observing microorganisms directly, this technique measures turbidity, which is the cloudiness that develops in a liquid culture as microbial cells multiply and scatter light.
Step 1: Preparing the liquid bacterial culture
The process begins with a bacterial suspension grown in a liquid nutrient medium. As microorganisms reproduce through binary fission, the culture becomes increasingly cloudy. This visible change in turbidity reflects the accumulation of microbial cells in the suspension and forms the basis for indirect growth measurement.
Step 2: Setting up the spectrophotometer
A small sample of the culture is placed into a clean cuvette, which is a transparent container designed for optical measurements. A control cuvette containing only sterile medium is also prepared. The spectrophotometer is then set to a specific wavelength of light, which must remain consistent throughout the experiment to ensure reliable comparison of readings.
Step 3: Passing light through the microbial suspension
The instrument directs a beam of light through the bacterial sample. As light passes through the suspension, microbial cells scatter and absorb part of the light energy. Because of this scattering effect, less light reaches the detector compared to the control sample. This reduction in transmitted light is directly related to the number of cells present.
Step 4: Measuring optical density or percent transmission
The spectrophotometer converts the detected light signal into numerical values such as percent transmission or absorbance, also known as optical density. As microbial growth increases, turbidity rises and optical density values increase accordingly. These measurements allow microbiologists to monitor population changes quantitatively over time.
Step 5: Interpreting results and recognizing limitations
Although spectrophotometry is rapid, non destructive, and suitable for real time monitoring of microbial growth, it does not distinguish between viable and nonviable cells. Dead cells can still scatter light and contribute to turbidity readings. For this reason, spectrophotometric analysis is often used together with viable counting methods such as plate counts to obtain a more complete understanding of microbial population dynamics.
Conclusion
Measuring microbial growth requires a combination of direct observation, statistical reasoning, and technological tools. Methods such as the Petroff Hausser chamber allow scientists to count total cells, while plate counts and CFU measurements focus on viable organisms. Serial dilution makes it possible to handle dense cultures, the Most Probable Number method provides estimates in complex samples, and spectrophotometry offers rapid indirect monitoring of population changes.
Together, these techniques enable microbiologists to quantify microbial populations accurately and apply this knowledge in clinical diagnosis, environmental monitoring, and industrial microbiology. Understanding these methods helps learners move beyond memorization and appreciate how laboratory reasoning is used to study organisms that are otherwise invisible to the naked eye.