Rabies pathogenesis describes the sequence of biological events that occur after infection with Rabies virus, the etiological agent of rabies. This virus belongs to the family Rhabdoviridae and is classified as an enveloped, negative sense single stranded RNA virus. Its bullet shaped structure and RNA dependent RNA polymerase are essential to its ability to replicate within host cells and spread through the nervous system.
Rabies is a zoonotic infection that is most commonly transmitted through the bite of an infected animal. After entering the body, the virus initially replicates in muscle tissue before gaining access to peripheral nerves. From there, it travels toward the central nervous system, where it causes progressive neurological disease. This unique pattern of neurotropic spread explains both the incubation period and the high fatality rate once clinical symptoms appear.
Understanding Rabies Pathogenesis requires examining viral structure, mode of transmission, neurotropism, replication strategy, immune evasion, and clinical progression. By analyzing each stage of infection, it becomes clear how early intervention through post exposure prophylaxis can prevent central nervous system involvement and alter the clinical outcome.

Etiological Agent and Viral Structure
The etiological agent of rabies is Rabies virus, a member of the family Rhabdoviridae. It is classified as an enveloped, negative sense single stranded RNA virus. Its structural organization directly determines how infection begins, how replication occurs, and how the virus spreads through nervous tissue. Understanding viral structure is essential for understanding Rabies Pathogenesis.
Enveloped Virion
Rabies virus is an enveloped virion surrounded by a lipid membrane derived from the host cell during viral budding. Embedded in this envelope are viral glycoproteins that function in receptor recognition and membrane fusion. The presence of an envelope makes the virus sensitive to environmental conditions such as drying and detergents. The envelope also plays a critical role in host cell entry by facilitating fusion with the host plasma membrane.
Negative Sense Single Stranded RNA Genome
The rabies genome consists of negative sense single stranded RNA. Because it is negative sense, it cannot be directly translated by host ribosomes. The viral RNA must first be transcribed into positive sense messenger RNA. This requirement distinguishes rabies from positive sense RNA viruses and explains why it must carry its own polymerase enzyme to initiate replication immediately after entry.
RNA Dependent RNA Polymerase
Rabies virus carries an RNA dependent RNA polymerase within the virion. This enzyme is essential because host cells do not possess enzymes capable of replicating RNA from an RNA template. The polymerase transcribes the negative sense genome into messenger RNA and also synthesizes new genomic RNA during replication. Without this enzyme, viral replication could not occur.
Bullet Shaped Capsid and Helical Nucleocapsid
Rabies virus has a distinctive bullet shaped capsid with a helical nucleocapsid structure. The nucleocapsid protects the viral RNA and ensures efficient packaging of the genome. The structural organization supports stability during transmission and facilitates controlled release of the genome once inside the host cell. The bullet shaped morphology is a defining feature of Rhabdoviridae and is important for viral assembly and budding.
Together, these structural features determine how Rabies virus attaches to host receptors, enters cells through membrane fusion, initiates RNA transcription, and establishes infection. Viral structure is therefore directly linked to the mechanisms of Rabies Pathogenesis.
Read Also: Clinical Diagnosis of Prions as Nontraditional Infectious Agents
Mode of Transmission of Rabies
Rabies Pathogenesis begins with exposure to the etiological agent, Rabies virus. The mode of transmission of rabies is primarily through the bite of an infected animal. Rabies is a zoonotic disease, meaning it is transmitted from animals to humans. Understanding transmission is essential because it explains how infection is established and why early intervention is critical.
Animal Reservoirs and Zoonotic Spread
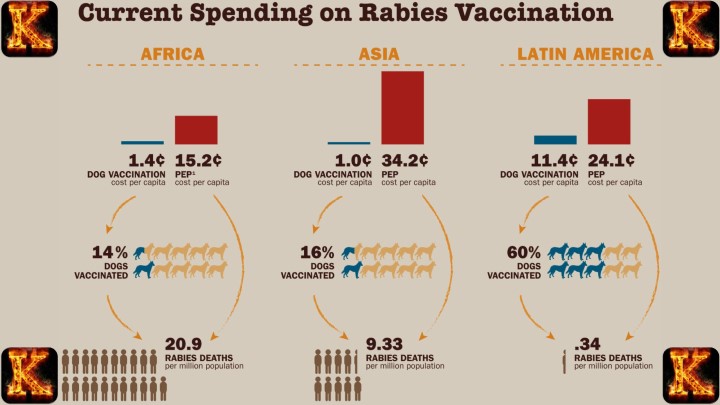
Dogs are the most common reservoir worldwide and account for the majority of human rabies cases globally. In the United States, wildlife reservoirs such as bats, raccoons, and foxes play a significant role in transmission. These animals carry the virus in their salivary glands, often without immediate visible symptoms. The presence of animal reservoirs maintains the virus in nature and allows continued spread to humans and other mammals.
Viral Entry Through Saliva
Rabies virus is present in infected saliva. During a bite, saliva containing virions is introduced directly into muscle tissue and sometimes subcutaneous tissue. This exposure bypasses many external immune barriers. The introduction of virus into tissue marks the first step in Rabies Pathogenesis. The virus does not immediately enter the bloodstream in high amounts, which is why early symptoms may be absent during the incubation period.
Initiation of Rabies Pathogenesis in Humans
After transmission, the virus begins local replication in skeletal muscle cells near the bite site. This early stage is often clinically silent, which contributes to delayed recognition. The pathogenesis of rabies virus at this stage depends on viral load, location of the bite, and host immune response. Once replication is established, the virus gains access to peripheral nerves, initiating neuroinvasion and progression toward the central nervous system.
Early Stage of Rabies Pathogenesis
The early stage of Rabies Pathogenesis begins immediately after the virus is introduced into muscle tissue through an animal bite. At this point, the virus is localized near the site of entry and has not yet reached the central nervous system. Understanding this stage is critical because it represents the window during which rabies virus treatment can successfully prevent severe disease.
Receptor Binding at the Neuromuscular Junction
After entering muscle tissue, Rabies virus binds to specific receptors at the neuromuscular junction. One important receptor is the nicotinic acetylcholine receptor. This receptor binding allows the virus to attach securely to host cells. The interaction between viral glycoproteins and host cell receptors is a key step in Rabies Pathogenesis because it determines tissue tropism and initiates infection at the cellular level.
Viral Entry Through Membrane Fusion
Once attached, the enveloped virus enters the host cell through membrane fusion. The viral envelope merges with the host cell membrane, allowing the viral nucleocapsid and negative sense single stranded RNA genome to enter the cytoplasm. Because the genome is negative sense RNA, the virus relies on its RNA dependent RNA polymerase to begin transcription. This step marks the start of intracellular replication.
Local Replication in Skeletal Muscle
Initial replication occurs locally within skeletal muscle cells. During this phase, viral RNA is transcribed and translated into viral proteins, and new virions are assembled. This localized replication explains the incubation period of rabies. The duration of incubation depends on factors such as the viral load and the anatomical location of the bite. Bites closer to the head often result in shorter incubation periods because the virus has a shorter distance to travel to the brain.
Clinical Significance of the Early Stage
The early stage of Rabies Pathogenesis is often asymptomatic, which can delay diagnosis. However, this stage is also when post exposure prophylaxis is most effective. Administration of human rabies immunoglobulin and rabies vaccination during this phase can neutralize the virus before it enters peripheral nerves. Once neuroinvasion begins, prevention becomes significantly more difficult.
Read Also: How Microscopy Helps Identify Bacteria in Clinical Samples
Neurotropism and Movement Along Peripheral Nerves
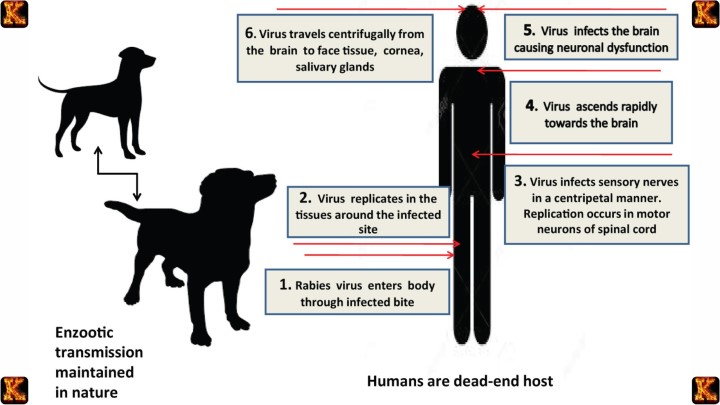
A defining characteristic of Rabies Pathogenesis is neurotropism, which refers to the virus having a strong preference for infecting nervous tissue. After initial replication in skeletal muscle, Rabies virus does not remain confined to the site of the bite. Instead, it specifically targets peripheral nerves, which allows it to move toward the central nervous system. This selective targeting of neural tissue explains the unique pathophysiology of rabies in humans.
Entry into Peripheral Nerves
Following local replication, the virus binds to receptors at the neuromuscular junction and gains entry into peripheral nerve endings. This step marks a major transition in Rabies Pathogenesis because the infection shifts from muscle cells to neurons. Once inside nerve cells, the virus becomes less accessible to circulating immune defenses, reducing the effectiveness of antibodies present in the bloodstream.
Retrograde Axonal Transport
Rabies virus travels retrograde along axons through a process known as axonal transport. This means the virus moves from the peripheral site of infection toward the spinal cord and brain. The movement is relatively slow but highly efficient. The distance between the bite site and the brain influences the incubation period. Bites closer to the head generally result in a shorter progression time because the virus has a shorter path to the central nervous system.
Immune Evasion and Disease Progression
Once inside neurons, Rabies virus is shielded from circulating antibodies because immune molecules have limited access to neural tissue. This immune evasion mechanism is central to Rabies Pathogenesis. As the virus ascends to the spinal cord and brain, it begins widespread replication in the central nervous system. At this stage, neurological symptoms emerge, and the clinical course becomes severe.
Pathophysiology of Rabies in Humans
In Rabies Pathogenesis, the most severe stage begins when Rabies virus reaches the central nervous system. After retrograde movement along peripheral nerves, the virus enters the spinal cord and brain, where viral replication increases rapidly. The infection of neurons leads to inflammation of brain tissue and the development of encephalitis. This neurological involvement defines the critical turning point in the progression of disease.
Viral Replication in the Brain
Once inside the brain, Rabies virus replicates within neurons using its RNA dependent RNA polymerase. The infection disrupts normal neuronal function rather than causing widespread cell destruction. This dysfunction alters neurotransmission and leads to abnormal neurological activity. The accumulation of viral particles and inflammatory responses contributes to the severe pathophysiology of rabies in humans.
Development of Encephalitis
Encephalitis results from viral replication combined with immune mediated inflammation. Brain swelling and impaired neural signaling lead to progressive neurological impairment. This stage of Rabies Pathogenesis is associated with rapid clinical deterioration. The central nervous system becomes the primary site of viral activity, and the immune system is unable to effectively eliminate the infection from neural tissue.
Rabies Virus Symptoms
Clinical manifestations reflect widespread neurological dysfunction. Early symptoms include fever and headache, followed by agitation and anxiety. Hydrophobia develops due to painful spasms of throat muscles during swallowing. As the disease progresses, paralysis, confusion, and altered consciousness occur. These rabies virus symptoms signal advanced central nervous system involvement and indicate a poor prognosis.
Centrifugal Spread to Salivary Glands
After central nervous system infection, the virus spreads centrifugally along peripheral nerves to other tissues, particularly the salivary glands. Viral replication in salivary tissue ensures that infectious virions are present in saliva, facilitating transmission to new hosts. This final stage completes the biological cycle of Rabies Pathogenesis and explains how the disease perpetuates within animal and human populations.
The Four Stages of Rabies
Incubation Stage
The incubation stage of rabies pathogenesis begins after the virus enters the body through an infected animal bite. Rabies virus is present in the saliva of the animal and enters the muscle tissue at the bite site. During this stage, the virus replicates locally in muscle cells without causing any noticeable symptoms. The duration of this stage can vary from a few days to several months depending on the proximity of the bite to the central nervous system and the amount of virus introduced. Early intervention during this stage can prevent the virus from reaching the nervous system.
Prodromal Stage
In the prodromal stage, the virus starts moving along peripheral nerves toward the central nervous system. Early symptoms appear and include fever, malaise, fatigue, and tingling or itching at the site of the bite. These symptoms are nonspecific but indicate that rabies pathogenesis is progressing. The virus is still spreading within the nervous system but has not yet caused severe neurological damage. Recognizing these signs is critical for timely post-exposure treatment to stop further viral progression.
Acute Neurologic Stage
During the acute neurologic stage, rabies virus reaches the brain and spinal cord. Patients begin to show neurological symptoms such as agitation, confusion, hydrophobia, anxiety, and sometimes hallucinations. Rabies pathogenesis is now in its most severe phase as the virus causes widespread damage to neurons. Once this stage begins, prognosis is extremely poor, and supportive care is the main option because antiviral treatment is largely ineffective at this point.
Coma and Death
The final stage of rabies pathogenesis is marked by progressive neurological deterioration leading to coma. The virus continues to damage the central nervous system, causing respiratory failure and death. This stage highlights why early detection and immediate post-exposure prophylaxis are crucial. Clinical rabies is almost universally fatal once the patient enters the acute neurologic stage.
Read Also: Risk Prediction Models in Clinical Practice
Fatality After Symptom Onset
1. Extensive Neuronal Damage
Once rabies virus reaches the central nervous system, it infects neurons throughout the brain and spinal cord. The virus replicates inside neurons without immediately destroying them, but this persistent infection gradually disrupts normal neural communication. Neuronal pathways controlling essential functions such as breathing, swallowing, and heart regulation are progressively impaired. This extensive neuronal damage is a major factor in the high fatality rate observed in clinical rabies. Even with supportive care, the loss of neural function cannot be reversed.
Rabies pathogenesis at this stage is marked by widespread infection of critical brain regions including the hippocampus, brainstem, and spinal motor neurons. The virus’s neurotropic nature allows it to spread efficiently along nerve fibers, leading to irreversible structural and functional damage. Early intervention before the virus enters neurons is therefore essential to prevent progression to this fatal stage.
2. Severe Brain Inflammation
Rabies pathogenesis also involves significant brain inflammation, which contributes to neurological deterioration. As the immune system recognizes the virus, inflammatory responses occur in the brain and surrounding tissues. This inflammation, or encephalitis, further damages neurons and disrupts normal brain signaling. Swelling within the confined space of the skull increases intracranial pressure, worsening symptoms such as agitation, confusion, and hydrophobia. Inflammation intensifies neuronal dysfunction and accelerates the progression toward coma.
Despite the immune response, the virus is largely shielded within neurons, limiting the effectiveness of the inflammatory reaction. The combination of viral replication and inflammation creates a vicious cycle of neural injury that cannot be reversed once clinical symptoms appear. Understanding this stage highlights why rabies pathogenesis is considered almost universally fatal after symptom onset and why prevention is more effective than treatment.
3. Limited Immune Clearance of Virus from Neurons
Another key factor in fatality during rabies pathogenesis is the immune system’s inability to eliminate the virus from neurons. Rabies virus resides inside neurons where it is largely hidden from antibodies and immune cells. Unlike infections in other tissues, the immune system cannot efficiently clear the virus from the nervous system. This allows continued viral replication and spread throughout the central nervous system.
Once the virus establishes itself in the brain, post-exposure prophylaxis or antiviral treatments are no longer effective. The inability of the immune system to control the virus explains why supportive and palliative care is the main option after neurological symptoms appear. Early vaccination and immediate wound care remain critical because the immune system is more effective before the virus enters neurons.
4. Supportive and Palliative Care
After the onset of clinical symptoms, rabies pathogenesis is largely irreversible, and treatment focuses on relieving suffering. Supportive care may include sedation, hydration, and management of seizures or respiratory difficulty, but it does not stop the virus from progressing. Palliative care ensures comfort in the final stages of the disease, addressing pain, agitation, and anxiety.
The high fatality rate at this stage underscores the importance of early intervention following animal bites. Rabies pathogenesis shows that once neurological involvement begins, there is almost no chance of recovery. Prevention through vaccination, avoiding exposure to infected animals, and prompt post-exposure prophylaxis remain the only reliable strategies to combat rabies.
Rabies Virus Treatment and Post Exposure Prophylaxis
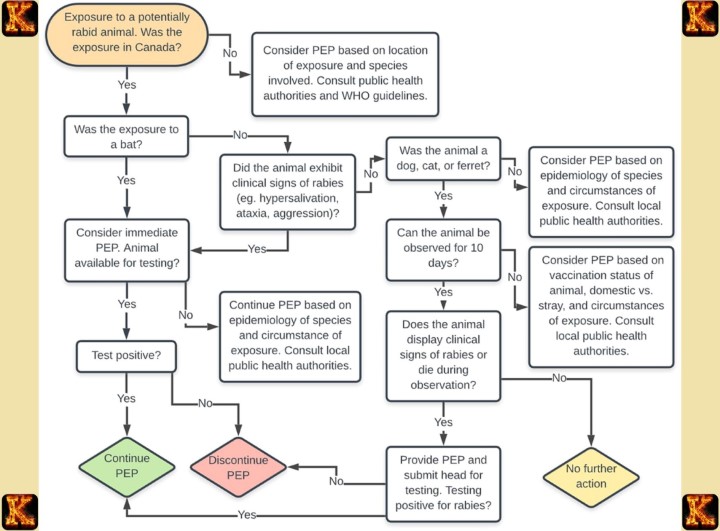
1. Timing of Treatment
A key concept in rabies pathogenesis is that treatment is effective only before the virus reaches the central nervous system. Rabies virus initially replicates at the bite site in muscle tissue and slowly travels along peripheral nerves toward the brain. This slow progression provides a critical window for medical intervention. Once the virus invades neurons and reaches the central nervous system, neurological symptoms appear, and rabies becomes almost universally fatal. Therefore, early recognition of exposure through animal bites is essential.
Immediate wound care, such as thorough washing with soap and water, reduces viral load at the entry site. The timing of treatment is crucial because post exposure prophylaxis relies on preventing the virus from establishing infection in the nervous system. Understanding the stages of rabies pathogenesis helps explain why delayed treatment after symptom onset is ineffective.
2. Human Rabies Immunoglobulin
Human rabies immunoglobulin provides passive immunity by neutralizing virus particles at the wound site. It contains pre-formed antibodies specific to rabies virus, which bind directly to virions and prevent their entry into peripheral nerves. This immediate protection is especially important in cases with high-risk exposure or deep bites from infected animals. Administering immunoglobulin as part of post exposure prophylaxis reduces viral replication at the local site and slows progression along the peripheral nervous system.
The effectiveness of this treatment is linked directly to rabies pathogenesis, as it targets the virus before neuronal invasion. Immunoglobulin is injected around the wound and sometimes systemically, offering rapid protection while the body prepares its own immune response. Without this intervention, the virus can move undetected into the nervous system, where it becomes resistant to treatment.
3. Rabies Vaccine Series
The rabies vaccine series stimulates active immunity by triggering the production of specific antibodies against rabies virus. Unlike immunoglobulin, the vaccine does not provide immediate protection, but it primes the immune system to respond effectively once viral particles enter the body. The immune system generates neutralizing antibodies that can recognize and eliminate the virus before it reaches the central nervous system. Post exposure prophylaxis typically involves multiple doses over several weeks, ensuring robust antibody levels.
The success of this strategy relies on the slow progression of rabies virus through peripheral nerves. Because the virus does not immediately invade the brain, the vaccine-induced immune response can intercept the infection during the incubation and prodromal stages of rabies pathogenesis. This highlights why vaccination after exposure is effective and why prevention remains critical in high-risk regions.
4. Importance of Post Exposure Prophylaxis
Post exposure prophylaxis is effective because rabies virus travels slowly through peripheral nerves before central nervous system invasion. This delay provides a crucial window for passive and active immunity to neutralize the virus. Combining human rabies immunoglobulin and the vaccine series ensures both immediate and long-term protection. The strategy directly targets the stages of rabies pathogenesis, preventing the virus from reaching neurons and causing irreversible neurological damage.
Early administration following a bite or scratch from a potentially infected animal significantly reduces the risk of fatality. Education on recognizing exposure, rapid wound care, and timely access to post exposure prophylaxis is essential for rabies prevention. Understanding this process demonstrates why rabies remains preventable even after exposure, unlike once symptoms of central nervous system involvement develop, where rabies pathogenesis makes recovery nearly impossible.
Read Also: Per Patient Day System in Tackling Nurse Burnout
How Rabies Works Biologically
1. Viral Neuroinvasion
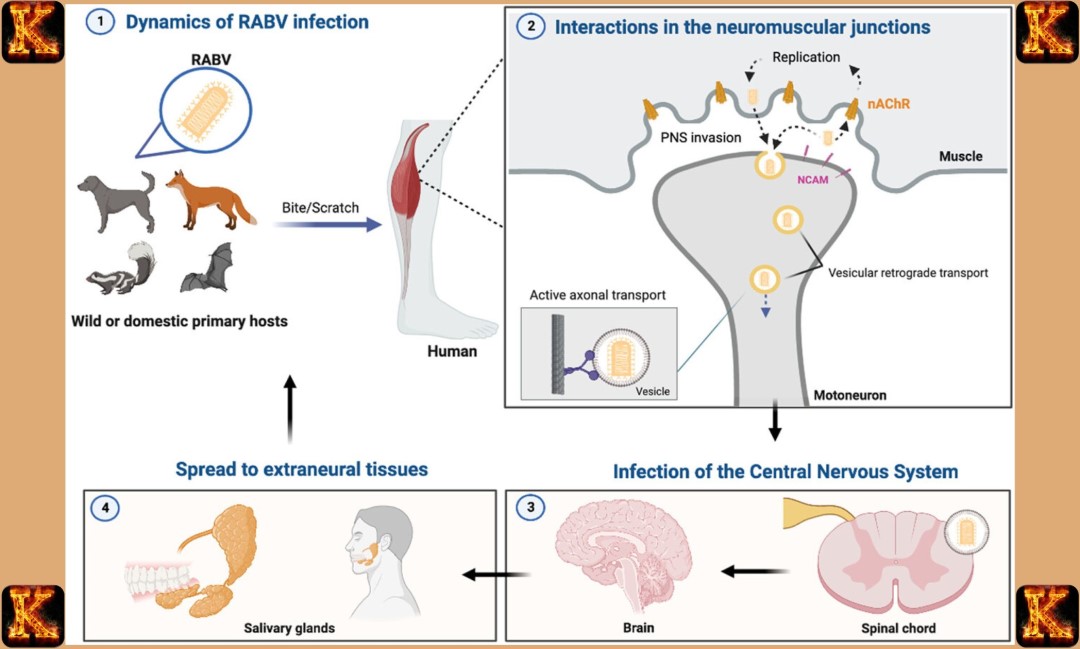
Rabies pathogenesis begins when virions from infected animal saliva enter muscle tissue during a bite. The virus initially replicates locally in the muscle without causing symptoms, marking the start of infection. From the muscle, rabies virus attaches to peripheral nerves and travels retrogradely toward the central nervous system. The virus specifically targets neurons, taking advantage of their long axons to move efficiently toward the spinal cord and brain. This neurotropic behavior allows the virus to evade early immune detection, as antibodies cannot readily access neurons.
Viral replication along these pathways eventually leads to infection of the brain, where widespread neuronal damage and inflammation occur. Understanding this stage highlights the critical importance of early intervention before the virus reaches neural tissue, as once neurons are infected, rabies pathogenesis progresses rapidly toward fatal neurological disease.
2. Host Immune Activation
Rabies pathogenesis involves a race between viral spread and the host immune response. If the immune system is activated early, through post-exposure prophylaxis including rabies immunoglobulin and vaccine administration, antibodies can neutralize the virus at the bite site. Human rabies immunoglobulin provides immediate passive immunity, binding to virions before they enter peripheral nerves. The rabies vaccine induces active immunity by stimulating antibody production, which takes days but ensures long-term protection.
Early immune activation can prevent the virus from reaching the central nervous system, effectively stopping rabies pathogenesis. Wound care, rapid medical attention, and timely prophylaxis are therefore essential, as the immune system alone cannot overcome the virus once it has invaded neurons. This illustrates how the timing of intervention determines whether rabies infection will be prevented or progress toward fatal outcomes.
3. Brain Infection and Fatal Encephalitis
If the rabies virus reaches the brain before effective immunity develops, rabies pathogenesis proceeds to severe neurological disease. Viral replication in the central nervous system leads to extensive neuronal damage and inflammation, resulting in encephalitis. This stage is marked by agitation, hydrophobia, paralysis, and other neurological symptoms. The immune system is largely unable to clear the virus from neurons, making clinical rabies almost universally fatal. Fatal encephalitis occurs because neurons are essential for controlling vital functions such as respiration and heart activity, and their damage cannot be reversed.
Once clinical symptoms appear, treatment is limited to supportive care, and recovery is exceedingly rare. Understanding this stage emphasizes why post-exposure prophylaxis must be administered immediately after exposure to prevent the virus from reaching the central nervous system and triggering this lethal phase of rabies pathogenesis.
4. Timing Determines Outcome
The outcome of rabies infection is determined by the timing of immune intervention relative to viral spread. Rabies pathogenesis illustrates a race between the virus traveling through peripheral nerves and the host generating protective antibodies. Rapid administration of human rabies immunoglobulin and the vaccine series allows the immune system to neutralize the virus before neuroinvasion. Delays in treatment allow the virus to reach the central nervous system, where neurological damage becomes irreversible.
This timing-dependent progression explains why rabies is preventable with early post-exposure prophylaxis but almost always fatal after symptom onset. Education on identifying exposures, immediate wound care, and prompt medical response is essential to prevent rabies from completing its pathogenesis cycle. Awareness of this biological mechanism reinforces the critical role of timing in determining clinical outcomes and highlights why prevention is the primary strategy against rabies.

Comments are closed!